Abstract
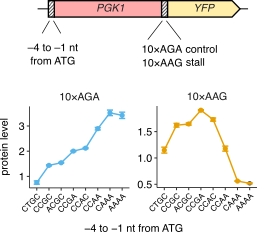
The canonical model of eukaryotic translation posits that efficient translation initiation increases protein expression and mRNA stability. Contrary to this model, we find that increasing initiation rate can decrease both protein expression and stability of certain mRNAs in the budding yeast Saccharomyces cerevisiae. These mRNAs encode a stretch of polybasic residues that cause ribosome stalling. Our computational modeling predicts that the observed decrease in gene expression at high initiation rates occurs when ribosome collisions at stalls stimulate abortive termination of the leading ribosome or cause endonucleolytic mRNA cleavage. Consistent with this prediction, the collision-associated quality-control factors Asc1 and Hel2 (orthologs of human RACK1 and ZNF598, respectively) decrease gene expression from stall-containing mRNAs only at high initiation rates. Remarkably, hundreds of S. cerevisiae mRNAs that contain ribosome stall sequences also exhibit lower translation efficiency. We propose that inefficient translation initiation allows these stall-containing endogenous mRNAs to escape collision-stimulated reduction in gene expression.