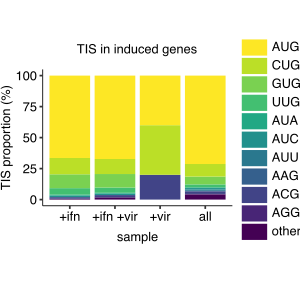Abstract
Translation can initiate at alternate, non-canonical start codons in response to stressful stimuli in mammalian cells. Recent studies suggest that viral infection and anti-viral responses alter sites of translation initiation, and in some cases, lead to production of novel immune epitopes. Here we systematically investigate the extent and impact of alternate translation initiation in cells infected with influenza virus. We perform evolutionary analyses that suggest selection against non-canonical initiation at CUG codons in influenza virus lineages that have adapted to mammalian hosts. We then use ribosome profiling with the initiation inhibitor lactimidomycin to experimentally delineate translation initiation sites in a human lung epithelial cell line infected with influenza virus. We identify several candidate sites of alternate initiation in influenza mRNAs, all of which occur at AUG codons that are downstream of canonical initiation codons. One of these candidate downstream start sites truncates 14 amino acids from the N-terminus of the N1 neuraminidase protein, resulting in loss of its cytoplasmic tail and a portion of the transmembrane domain. This truncated neuraminidase protein is expressed on the cell surface during influenza virus infection, is enzymatically active, and is conserved in most N1 viral lineages. We do not detect globally higher levels of alternate translation initiation on host transcripts upon influenza infection or during the anti-viral response, but the subset of host transcripts induced by the anti-viral response is enriched for alternate initiation sites. Together, our results systematically map the landscape of translation initiation during influenza virus infection, and shed light on the evolutionary forces shaping this landscape.